What’s Up With Pharma in 2024? (LogiPharma Edition)

PAXAFE just wrapped its fourth LogiPharma Boston conference just two years after attending its first ever conference (also LogiPharma Boston)!
Ahh to be young again…

Two years ago, we were one of two booths (out of what I guess to be around ~100 exhibitors) that were leading the charge talking about data contextualization, predictive analytics and risk management.
While all of the standard conference talks were about ‘visibility,’ ‘track & trace’ ‘custom reporting’ and ‘intervention,’ we knew that visibility was just a stepping stone [albeit an important enabler] to the desired outcome for customers.
As such, we were already building our AI-enabled recommendation platform which would eventually become ATHENA — the nirvana state for customers where they could automate root cause, understand correlations in their network’s performance and take immediate actions on prescriptive recommendations backed by data-based evidence that the platform auto-generates.

Now, just two years later — the market’s messaging has caught up brought about by customer demand. We’d guess that over 30% of exhibitors had some iteration of ‘risk,’ ‘prediction’ or ‘context’ as a part of their messaging.
And now, once again, PAXAFE is leading the pack, showcasing the intended nirvana state of cold-chain logistics: being able to put your transportation operations on auto-pilot through prescriptive recommendation.
This wasn’t by accident. As I routinely share with employees, customers and investors that I speak with, this was Ashok and I’s vision from the earliest of days.
Except we knew that you could not build an accurate and consistent recommendation engine without first building the context, risk and predictive infrastructure that currently makes up our CONTXT intelligence platform.
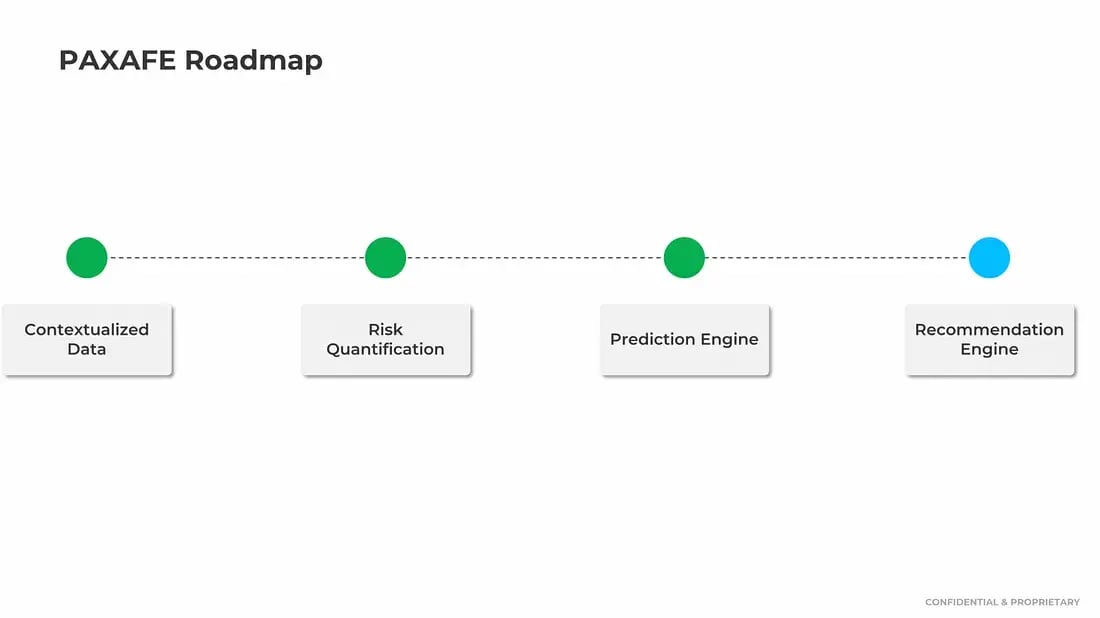
And so now, once again, after another successful LogiPharma conference, I’m sharing my top takeaways around what’s happening in the world of Pharma & Cold Chain Logistics!
ONE: IoT adoption is quick, but rollouts are slow

Ok, that’s kind of an oxymoron — I get it.
Pharma companies are making swift decisions around justifying the value of IoT and visibility and deciding to move forward with deployment.
That being said, when it comes time to actually deploy the devices, there are all sorts of roadblocks:
- Change management: there’s buy-in at a corporate level or at a senior level, but regional leaders / operations are not empowered or incentivized to ensure local success of rollout
- Device issues: devices are on their best behavior during the validation phase, but once you start scaling to thousands of devices, ping rate consistency, signal strength and battery reliability become problems
- Lack of KPIs or objectives in knowing what visibility will be solving for. It doesn’t always have to be that IoT needs to solve for product loss. Sometimes, it’s solely about having the data to make quicker, smarter network decisions for the future, and that’s OK.
TWO: There are still two different schools of thought in terms of consolidation vs. separation of powers

Some pharmaceutical companies prefer to procure our agnostic capabilities through channel partners that they’re already working with. This drives consolidation of vendor base and provides ‘one less partner’ to manage / work with.
Others want to have a complete separation of powers so that they’re not tethered to any one service provider — whether it be a 3PL, and IoT device provider or a packaging provider.
Both options come with their pros and cons.
THREE: Visibility Objectives are Fragmented

We spoke to a Pharma leader that told us that he doesn’t really care about saving ‘onesie twosie’ shipments — he cares about what actions he can take to prevent future losses en masse. Or reducing CO2 footprint. Or optimizing packaging performance. Or having the confidence to convert Air shipments to Ocean shipments.
We spoke to another Pharma leader that, upon seeing PAXAFE’s dynamic Lane Risk Assessment (LRA) tool (which helps do all of those aforementioned things!), said — “yeah, but how’s that going to help me intervene with problematic live shipments?”
These two individuals were from the same company.
We’re finding that functionally speaking, Quality, Transportation / Logistics and Supply Chain Planning care about fundamentally different things. And there’s a good chance that they have limited interaction with each other internally.
Secondarily, we’re also finding that more senior leaders are caring more and more about big ticket savings opportunities (forward looking), while mid-level leaders are more prone to focus on the here and now (live shipments).
And if you ask 3 different people from the same company as to why their company is investing in Visibility, it’s likely that you’ll get 3 different answers.
FOUR: Renewed Interest to Digitize Passive Data

For decades, temperature data loggers were the unsung heroes for quality leaders trying to determine whether a product should be accepted, rejected or quarantined.
Once real-time IoT sensors are fully vetted and their platforms are GxP validated, passive loggers are no longer needed.
That being said, the slow rollout and constant hurdles of IoT deployments (mentioned in point 1) is giving reason for Pharma to continue investing in Passive data.
Today’s problem with passive data lies in:
- No centralized repository for all passive data, with different Pharma regions having different SOPs around passive data collection & retention
- Manual product release process (for product approval, rejection)
- No passive lane insights at scale — limited understanding towards the performance of passive lanes
- Expensive middleware from sensor providers that are specific to one provider — making it more challenging and expensive for Pharma companies using 2 or 3 manufacturers
Pharma is coming to the conclusion that the majority of their lanes are still passive, and that they can make use of all of this data to establish some performance baselines, using it as a stepping stone to understand which lanes to prioritize with real-time IoT deployments.
FIVE: There’s an Opportunity to Improve the TRUST LAYER between Pharma and IoT
Pharma is placing some of the blame on delayed IoT rollouts attributed to lackluster device performance and device challenges, and then poor intervention services when temperature excursions get missed / are not actionable.
On the flip side, device providers are attributing many of the challenges towards over-promised demand and poor change management.
The reality is that there’s a little of both at play, and there’s an opportunity to build trust through flawless execution.
This means device providers have to be transparent and proactive when their devices are malfunctioning. And Pharma companies must adhere to financial commits and projections associated with inventory deployment.
Six: 🤑 Monitoring is too Expensive 🤑

Monitoring services teams are charging Pharma anywhere from $10/shipment all the way up to $200/shipment, depending on the level monitoring and intervention / action that is required.
This makes sense — having one person oversee the monitoring of hundreds of shipments per day… 15% of which will see some form of device alert and require some level of investigation… is not an insignificant task.
Pharma is seeking to understand how technology can augment productivity, and enable each monitoring agent to reduce device alert noise and only truly respond to alerts that pose a real risk to the product.
Seven: Logistics Service Providers (LSPs) are looking for ways to add more value and defend visibility’s ROI
There is still a race towards a cell-enabled hardware label that’s very much happening — the same race I’d mentioned back in 2022.
But until cost of components and hardware / firmware performance reach a state of feasibility, device providers are starting to look at other ways to add value.
This can include aggregate insights, packaging optimization consulting, custom reporting, monitoring & intervention services, etc.
As such, many of these LSPs are going through multiple decision points:
- Do we build versus buy?
- If we build, does that mean we need to materially change our hiring strategy?
- Do we verticalize for Pharma, who has unique requirements and workflows? Or do we implement something more generic for all of our customer segments?
- Are we going to cannibalize our existing services, or enhance them?
- Are we going to push towards more predictable software revenue, or find ways to charge transactionally?
That said, merely having a solution is one part of the equation. The more difficult part of the equation is aligning on the value quantification.
Pharma needs to have baseline data that already quantifies and establishes some of the following:
- full cost of a temperature excursion
- full cost of a delayed shipment
- time and cost associated with product release
- time and cost associated with validating a lane
- time and cost associated with packaging validation / simulation
- time and cost associated with IoT device validation / selection
- cost associated with route non-conformance
- inventory / turn cost associated with each incremental day that’s a part of a contracted lead time parameter
- time and cost associated with making a major Lane SOP parameter change (e.g. switching from air to ocean on a particular lane)
- Many others
In many cases, Pharma does not have this readily available — which makes value discussions more challenging for providers.
Eight: GenAI Doubted, But Shows Promise When Backed By Evidence

We hear doubt around the promise of GenAI after someone goes into ChatGPT, uploads some data and asks a few questions like “where is my risk today?” or “show me my delayed shipments.”
LLMs have no inherent comprehension of risk, delayed shipments or what’s important to you within the context of your cold chain transportation network. It doesn’t know what’s ‘normal,’ what your SOPs are, how you address or react to risk, and so forth.
To truly understand what’s possible, companies need access to a tuned model that understand the intersections of risk, decision intelligence, custom workflows, transportation and cold chain requirements — that’s been built on an underlying risk management infrastructure.
They’ll also need access to A LOT of training data.




